What is silicones? Silicones are synthetic polymers composed primarily of silicon and oxygen atoms arranged in repeating chains called siloxanes. These materials combine inorganic and organic chemistry characteristics, producing substances that remain stable under heat, moisture, and chemical exposure. Because of these properties, silicones are widely used in manufacturing, medical devices, sealants, electronics, and personal care products.
Key Takeaways
- They are synthetic compounds built from a structure containing silicon, oxygen, carbon, and hydrogen.
- Their backbone structure consists of repeating siloxane (Si–O–Si) bonds.
- Major types include silicone fluids, elastomers, and resins.
- Silicones differ from silicon, which is a natural chemical element.
- Industries like electronics, construction, medicine, and personal care frequently rely on these materials.
What Is Silicones in Chemistry?
In chemistry, what is silicones refers to a group of polymeric compounds formed by repeating siloxane (Si–O–Si) units with attached organic groups such as methyl or phenyl.
Key characteristics
- Backbone structure: alternating silicon–oxygen bonds
- Organic side groups: typically methyl (–CH₃)
- Polymer form: linear, cyclic, or cross-linked networks
Chemical representation
| Property | Description |
| Chemical backbone | –Si–O–Si– |
| Common substituents | Methyl, phenyl, vinyl |
| Polymer class | Organosilicon polymer |
Silicones behave differently from traditional carbon-based plastics because their silicon–oxygen bonds provide greater flexibility and thermal stability.
What Is Silicones Made Of?
Understanding what is silicones made of requires examining its elemental composition.
Silicones are produced from:
- Silicon – derived from silica sand
- Oxygen – forms the siloxane backbone
- Carbon and hydrogen – present in organic side groups
- Optional additives – fillers, catalysts, or stabilizers
Simplified production process
- Silicon is obtained from quartz or sand.
- Silicon reacts with methyl chloride to form organosilicon intermediates.
- Hydrolysis and polymerization create silicone polymers.
It generates materials known for their strong resistance to ultraviolet rays, oxidation, and temperature extremes.
What Are the Main Types of Silicones?
When discussing what is silicones, classification is important because different forms serve different industries.
Common silicone types
| Type | Structure | Typical Uses |
| Silicone fluids | Linear polymers | Lubricants, cosmetics |
| Silicone elastomers | Cross-linked rubber-like materials | Medical devices, seals |
| Silicone resins | Highly cross-linked networks | Coatings, electrical insulation |
These forms vary in viscosity, elasticity, and durability.
What Is the Difference Between Silicone and Silicon?
Many learners confuse these two terms when asking what is silicones.
| Feature | Silicon | Silicone |
| Chemical type | Element | Synthetic polymer |
| Periodic table | Yes | No |
| Composition | Pure silicon atom | Silicon + oxygen polymer |
| Uses | Semiconductors, solar panels | Sealants, medical tubing |
While silicon is a naturally found element, silicone is a manufactured polymer formed from silicon compounds.
What Are Silicone Polymer Examples?
Understanding what is silicones also involves recognizing common silicone polymer examples used in industry.
Examples include:
- Polydimethylsiloxane (PDMS) – widely used in cosmetics and lubricants
- Methyl silicone resins – protective coatings for electronics
- Silicone rubber – seals, gaskets, and medical tubing
- Fluorosilicone elastomers – fuel-resistant aerospace materials
These polymers are valued for flexibility, insulation, and resistance to extreme temperatures.
A brief note for learners: silicones are often discussed alongside related topics such as silicone rubber, silicone oil, and siloxane polymers in industrial chemistry references.
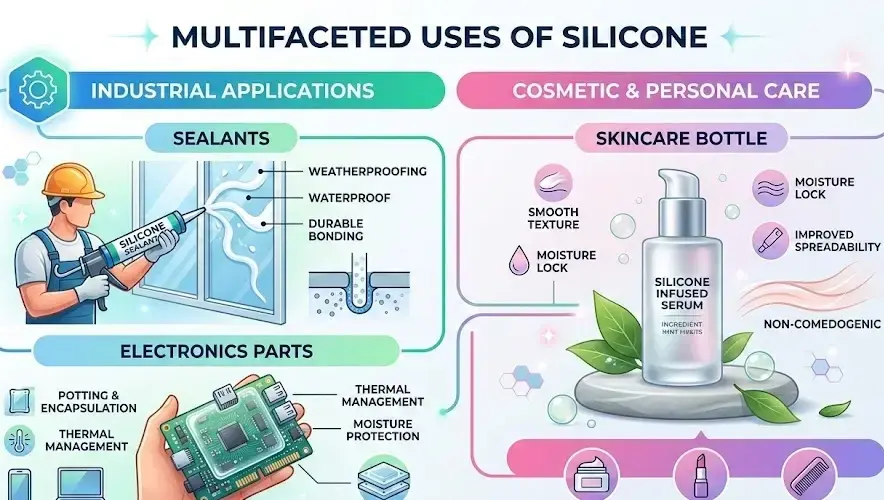
How Are Silicones Used Across Industries?
To fully understand what is silicones, examining practical applications is essential.
Major industrial uses
- Electronics: insulating materials and protective coatings
- Construction: sealants, adhesives, waterproofing
- Healthcare: implants, tubing, medical adhesives
- Automotive: gaskets, lubricants, thermal insulation
- Personal care: conditioners, primers, and skin protectants
These applications rely on silicones’ ability to resist heat, moisture, and chemical degradation.
Conclusion
Silicones are organosilicon polymers noted for their ability to withstand high temperatures, remain flexible, and resist chemical reactions. Their classification into fluids, elastomers, and resins explains their wide industrial use. Understanding this framework also clarifies their applications in specialized sectors, including personal care products discussed in Uses of silicone in cosmetics.
FAQ
What are silicones in skincare?
In cosmetic formulations, silicones act as synthetic compounds that provide a smooth surface layer while helping the skin hold moisture.
What is the definition of silicon?
Silicon is a chemical element (Si) in the periodic table widely used in electronics, semiconductors, and solar technology.
What are the three types of silicon?
The three common forms of silicon are crystalline silicon, amorphous silicon, and polycrystalline silicon.
What are 5 uses for silicon?
Silicon is used in semiconductors, solar panels, glass manufacturing, aluminum alloys, and electronic circuits.
Sources
https://www.elkem.com/products/silicones/silicone-polymers/
https://www.essentialchemicalindustry.org/polymers/silicones.html
https://www.simtec-silicone.com/blogs/how-is-silicone-produced/
https://www.shaalaa.com/question-bank-solutions/what-are-silicones_173850
https://www.americanchemistry.com/chemistry-in-america/chemistries/silicones
https://romakksilicones.com/what-is-silicone/
https://prototek.com/article/what-is-silicone/
https://www.shinetsusilicone-global.com/info/begin.shtml
https://byjus.com/jee/silicones/




